Iron and Steel Industry
(The Backbone of Industrial Civilization)
Let’s start with a fundamental question—why is iron and steel so important?
Just as a skeleton supports the human body, iron and steel form the framework of industries. Be it railways, bridges, factories, or skyscrapers—steel is the basic ingredient. That’s why it’s called the harbinger of the Industrial Revolution. The 18th and early 19th centuries saw the first wave of mechanisation, and steel was the lifeblood of that transformation.
Fast forward to the 21st century—steel is now a symbol of globalisation. Why? Because steel-producing countries are connected through trade, technology exchange, and capital investments. For example, when India builds a metro system with imported technology and steel from Japan, we see how globalisation operates through the veins of the steel industry.
Steel as an Economic Indicator
Now, how do we assess a country’s economic health? Just like doctors check your pulse or temperature, economists look at the per capita consumption of iron and steel. It shows how industrialised a country is.
High steel consumption = more construction, more manufacturing, more jobs = better economy.
So, when we say that India’s steel consumption is rising, it’s not just about factories—it’s about highways, homes, and even your metro rail.
India’s First Steps in Steelmaking
Let’s take a brief walk through history.
- First Iron and Steel Unit: It was established in Porto-Novo, Tamil Nadu. However, this was more like an experimental beginning—important, but not very impactful on a national scale.
- The real game changer came in 1907, when Jamshedji Tata established TISCO (Tata Iron and Steel Company) in Jamshedpur. This was the first truly modern steel plant in India. It wasn’t just an industrial move—it was a bold political message that India could be self-reliant even under British rule.
Steel Production Processes
Now let’s understand how steel is actually made. Think of these as different kitchens using different recipes to cook the same dish—steel 😊
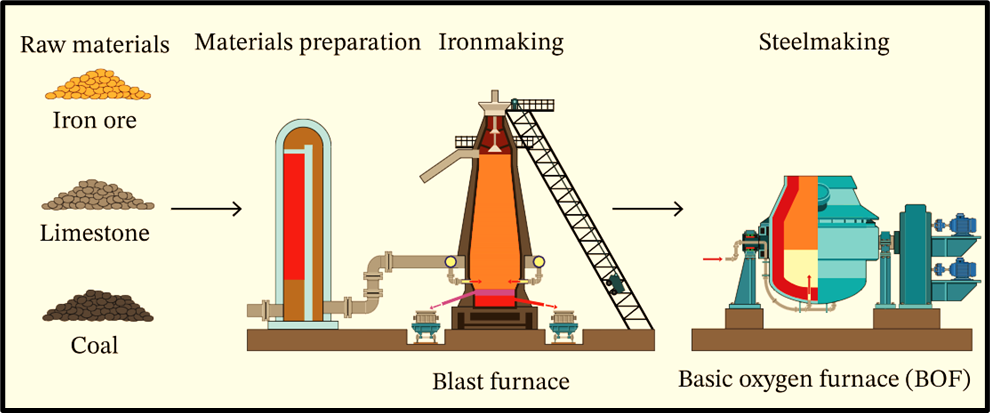
1. Blast Furnace–Basic Oxygen Furnace (BF–BOF) Route
This is the traditional and most widely used method, especially among large producers.
- Blast Furnace: This is where iron ore is converted into hot metal or pig iron. Pig iron is the solidified form, while hot metal is liquid. This is like boiling raw rice before making biryani—it prepares the base.
- Basic Oxygen Furnace: The hot metal is then refined into steel. Oxygen is blown into the furnace to remove impurities like carbon. It’s energy-intensive but produces high-quality steel.
Most large steel plants in the world—from Japan to the US—use this method.
2. Electric Arc Furnace (EAF): Recycling the Old
Think of this method like recycling old paper to make new notebooks.
- EAF uses 100% scrap metal as the raw material, plus lime and dolomite (which help in removing impurities).
- It consumes less energy compared to the BF-BOF route and is environmentally friendly.
That’s why many modern plants are shifting towards this route, especially in countries where scrap is abundant.
3. Induction Furnace (IF): The Budget-Friendly Kitchen 😊
Induction Furnace is the most cost-effective method, like cooking instant noodles instead of a full-course meal 😊
- Initially used only for stainless steel scrap, IFs began producing mild steel from the 1980s.
- It’s cheap and fast, but has a trade-off—it doesn’t refine the steel well. So the quality may not match BOF or EAF outputs. Yet, for small-scale production, it’s extremely useful.
To sum it up, the Iron and Steel Industry is not just an industry—it’s an indicator of where a country stands on the path of development. Whether it’s the skyscrapers you admire, the roads you drive on, or the mobile towers that connect us—it all begins in a steel plant.
In the next part, we’ll dive in technical details of the Iron and Steel Industry manufacturing.
Technical Analysis of Iron and Steel Industry
Before going into the technical details, let me clarify some basic concepts. See, we often hear that the iron and steel industry is a basic industry. What does that mean?
Think of it like the mother in a household. Every other industry—be it automobile, construction, shipbuilding, or even railways—depends on steel to function. That’s why it’s called basic.
It is also a heavy industry, because:
- It uses bulky raw materials like iron ore, coal (coke), and limestone.
- Its products (steel, pig iron) are heavy and difficult to transport.
So, both inputs and outputs are weighty—hence, heavy industry.
How Iron Becomes Steel: A Step-by-Step Journey
Smelting: Extracting Iron from Its Ore
Smelting is like the ancient process of extracting butter from milk—you heat, stir, and separate impurities.
- Iron ore is heated in a blast furnace.
- It’s mixed with coke (which acts as a fuel and reducing agent) and limestone (which removes impurities).
- The molten iron formed is cooled and solidified to make pig iron.
Pig iron is the raw, unrefined form of iron. It’s like wheat before it becomes bread. To convert it into steel, strengthening elements like manganese are added.
Understanding Impurities: Friends and Foes
Just like in a classroom—some students are helpful in small doses; others are disruptive no matter what—impurities in iron ore behave similarly 😊
Harmful Impurities
- Sulphur:
- Most dangerous.
- Forms iron sulphide, which is brittle and weakens steel.
- Tin:
- Forms a brittle film, makes steel practically useless.
- Oxygen:
- Forms oxides that reduce steel’s strength.
Beneficial (in Small Quantities)
- Silicon:
- Increases hardness slightly, helps remove oxygen.
- Lead:
- Improves machinability (easier to cut/shape), increases tool life.
- Manganese:
- Acts as a deoxidant, neutralizes sulphur’s bad effects.
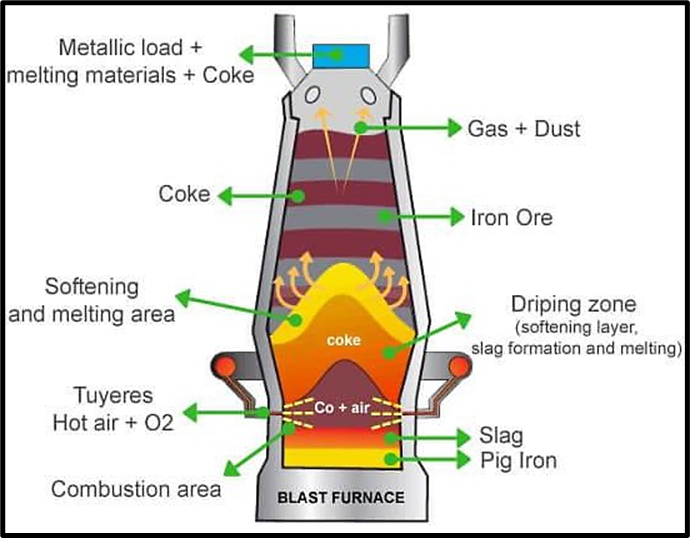
What Happens Inside a Blast Furnace?
Imagine a huge vertical oven where three ingredients go in:
- Iron ore
- Coke (fuel and reducing agent)
- Limestone (flux)
A hot blast of air is blown in from the bottom, and this sets off a series of chemical reactions.
Products of the Blast Furnace:
- Liquid Iron (Pig Iron)
- Slag (waste material of melted impurities)
- Gases like CO and CO₂
Why Use Coke Instead of Coal?
Coal is like raw rice—needs to be processed before use. Coke is the processed version of coal with:
- Fewer impurities
- Higher carbon content (~90%)
- Sulphur is removed, which is otherwise harmful
So, coke is preferred for its cleaner, stronger, and more efficient performance in melting iron.
Reduction: The Science Behind Iron Extraction
The term reduction here means removing oxygen from iron oxides which happens through a series of chemical reactions:
- 3Fe2O3 + CO ⟶ CO2 + 2Fe3O4
- Fe3O4 + CO ⟶ CO2 + 3FeO
- FeO + CO ⟶ CO2 + Fe (pure metal)
Let’s simplify the chemical reactions:
- Fe₂O₃ → Fe₃O₄
- Fe₃O₄ → FeO
- FeO → Fe (pure iron)
The hero here is carbon monoxide (CO)—it grabs the oxygen from iron oxides and takes it away as CO₂. But yes, these are pollutants, so modern steelmaking must find cleaner alternatives.
Is Electric Smelting a Better Option?
Yes—and no.
Electric smelting is a clean alternative using electricity instead of coke. But there are challenges:
- Huge infrastructure overhaul required
- Expensive to switch from coke to electric
- Electricity itself is often made from coal, especially in India
However, where hydroelectric power is cheap and available, electric smelting is viable. A good Indian example is:
👉 Visvesvaraya Iron and Steel Plant in Bhadravathi (Karnataka)
It uses electricity from Sharavathi hydro power plant near Jog Falls.
Role of Limestone: The Hidden Cleanser
Limestone is like the detergent in the washing machine—it removes stubborn dirt.
- It breaks down to CaO (Calcium oxide) in heat.
- CaO reacts with sulphur and forms slag.
- This slag collects impurities like silica and alumina.
Slag, being lighter, floats above molten iron and can be separated easily.
To Summarize:
The iron and steel industry is basic, heavy, and essential. The production process involves:
- Careful removal of harmful impurities
- Smart use of chemical reactions to extract pure metal
- Coke as the primary fuel (despite pollution) due to its efficiency
- Possibility of electric alternatives where conditions allow
- Importance of limestone in cleaning and refining the product
All of this shows why steelmaking is both a science and an art, blending chemistry, engineering, and economy.
Types of Iron and Steel: Composition, Properties, and Applications
Pig Iron: The First Avatar of Iron
Think of Pig Iron as the crude oil of the iron world—it’s not the end product, but it’s the foundation for everything else.
- It is the intermediate product obtained from smelting iron ore in a blast furnace.
- It contains iron as the major component (93–95%), but also has impurities or minor elements like:
- Carbon (4.1–4.4%) – This is the most significant. High carbon makes it hard and brittle.
- Silicon, sulphur, phosphorus, manganese – Present in small but impactful quantities.
📌 Key Insight: The carbon content in pig iron is quite high. And this high carbon is the reason why pig iron is hard but brittle, meaning it can break easily under stress.
Cast Iron: The Tough but Brittle Cousin
Now imagine you tweak pig iron just a bit—you get Cast Iron.
- Carbon content is above 2%, often between 2.5–4%.
- It also has silicon as a major alloying element.
- Due to this composition, cast iron is brittle—strong in compression but weak in tension.
🛠️ Applications:
- Engine blocks
- Pipes
- Cookware like cast iron pans
You can think of cast iron like a strong but rigid person—great at holding weight, but not flexible or adaptable.
Steel: The Balanced and Versatile Metal
When you reduce the carbon content to less than 2.1%, you get Steel.
This makes all the difference:
- Stronger yet more ductile than cast iron.
- Can be rolled, shaped, and welded.
- Its properties can be tailored by slightly varying carbon content and adding alloying elements.
It’s like having a well-trained athlete—strong, flexible, and adaptable.
Stainless Steel: The Gentleman of Steels
This is steel with polish and manners—resists rusting, corroding, and staining.
Composition:
- Minimum 10.5% chromium (forms a protective oxide layer)
- Often includes nickel, manganese, molybdenum, etc.
📌 Why is chromium important?
Chromium forms a thin, stable film of chromium oxide on the surface. This film acts like a shield against water, oxygen, and acids.
🛠️ Applications:
- Kitchen utensils
- Surgical tools
- Architecture
- Food processing industries
Wrought Iron: The Sculptor’s Delight
Wrought iron is almost the opposite of cast iron.
- It has very low carbon content.
- Made by mixing molten iron with slag, which gives it a fibrous structure.
- It is soft, malleable, and corrosion-resistant.
📌 Key Feature: You can heat and reshape it—perfect for ornamental gates, furniture, and historical railings.
Think of wrought iron like wet clay—easy to mould, not very strong, but long-lasting and doesn’t “rot” easily.
To Summarize: The Forms of Iron at a Glance
| Type | Carbon Content | Strength | Brittleness | Rust Resistance | Uses |
|---|---|---|---|---|---|
| Pig Iron | 4.1–4.4% | Hard | Very brittle | Low | Raw material for steel, cast iron |
| Cast Iron | >2% | Strong (compressive) | Brittle | Low | Engines, pans, pipes |
| Steel | ≤2.1% | Strong & ductile | Balanced | Moderate | Construction, tools, machines |
| Stainless Steel | ≤2.1% + 10.5% Chromium | Corrosion-resistant | Ductile | High | Utensils, surgical instruments |
| Wrought Iron | Very low | Malleable | Not brittle | High | Railings, furniture, artistic work |
Just by tuning the carbon content and adding a few more elements, we create a whole spectrum of materials—from the rigid cast iron to the elegant stainless steel.
And that’s the beauty of metallurgy—you start with one metal (iron) and end up with many personalities suited to different jobs.
In the next section we move on to the Locational Factors of Iron and Steel Industry.
